When you pick up a prescription, you might notice your pill looks different than last time. Maybe it’s a different color, shape, or has a weird marking. That’s not a mistake - it’s probably a generic drug. But how do you know it’s real and safe? With counterfeit drugs on the rise, it’s not enough to trust the pharmacist. You need to know what to look for - and what to avoid.
What Makes a Generic Drug Legitimate?
A legitimate generic drug isn’t just a cheaper copy. It’s a medically identical version of a brand-name drug, approved by strict regulators like the FDA, EMA, or Health Canada. The key? It must contain the exact same active ingredient, in the same strength, and work the same way in your body. That’s not marketing speak - it’s science. The FDA requires generics to prove they’re bioequivalent: meaning your body absorbs them within 80% to 125% of the brand-name version. A 2021 study in JAMA Internal Medicine analyzed over 2,000 approved generics and found 98.7% met this standard. The average difference in absorption was less than 1% - practically identical.Why Do Generics Look Different?
You might think a different color means a fake. But that’s not true. Legitimate generics can look completely different from the brand-name version. Why? Because trademark laws protect the appearance of brand-name drugs. So, generic manufacturers can’t copy the shape, color, or logo. A blue oval pill from the brand might become a white oval from the generic. A red capsule might turn into a green one. That’s legal. That’s normal. What’s not normal? A pill that looks cracked, crumbly, or has a weird texture. If it feels dusty, sticky, or smells odd, that’s a red flag.What to Check on the Packaging
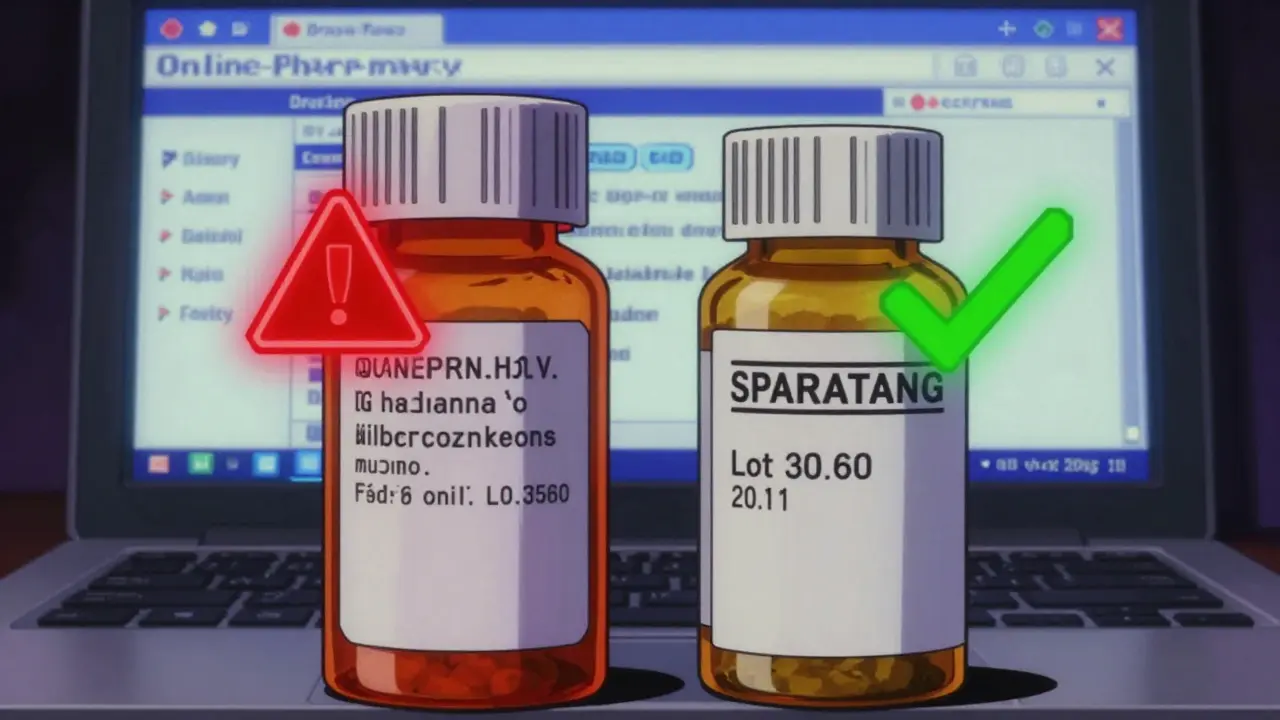
Legitimate generics come in properly labeled containers. Look for these details:- The drug name (like “amlodipine”) and strength (like “5 mg”)
- The manufacturer’s name - not just a generic label like “Pharmacy RX”
- A lot number and expiration date
- A barcode or printed code that matches the pharmacy’s records
Check the Pharmacy - Not Just the Pill
Most counterfeit drugs come from fake online pharmacies. The National Association of Boards of Pharmacy (NABP) says 96% of websites selling drugs without a prescription are illegal. So, where you buy matters as much as what you buy.- Use only pharmacies with a .pharmacy domain. You can verify this at nabp.net - it takes less than two minutes.
- Make sure the pharmacy requires a valid prescription. No pharmacy legally sells controlled medications without one.
- If you’re buying online, check if they’re listed in the Verified Internet Pharmacy Practice Sites (VIPPS) program. As of late 2023, only 62 online pharmacies in the U.S. held this accreditation.
Use the FDA’s Orange Book
The FDA’s Orange Book is a public database that lists all approved generic drugs and their brand-name equivalents. As of September 2023, it included over 14,800 generic products. You can search it online by drug name or manufacturer. If the generic you’re taking isn’t listed, it’s not approved - period. This isn’t just for doctors. Consumers can use it too. If your pill’s manufacturer is listed and matches the lot number on your bottle, you’re likely safe.What to Do If Something Feels Off
Sometimes, a generic might work differently - not because it’s fake, but because your body reacts to inactive ingredients. That’s rare, but it happens. If you’ve been taking a generic for months and suddenly feel worse, notice new side effects, or the medication seems less effective, don’t assume it’s counterfeit. But don’t ignore it either. Here’s what to do:- Compare the pill to your last refill. Did the color, shape, or imprint change? If yes, ask your pharmacist why.
- Check the lot number on the bottle. Go to the FDA’s Drug Recall Database and search it. If there’s a recall, you’ll know immediately.
- Call the manufacturer. Most big generics like Teva, Sandoz, or Mylan have customer service lines. Ask them to confirm the lot number matches their production.
- If you suspect fraud, report it to the FDA’s MedWatch program. In 2022, over 1,200 counterfeit drug reports came in - many from patients who noticed unusual aftertastes or sudden loss of effect.

Real Stories: What People Have Seen
On Reddit’s r/pharmacy, users shared stories. One person took a generic version of their blood pressure pill for years. Then, the color changed from white to yellow. They were worried - until they called the pharmacy. The pharmacist showed them the new manufacturer’s label. The pill was still approved. The user kept taking it - and stayed healthy. Another person bought a generic erectile dysfunction pill from a website that looked professional. The pills were the right color, had the right imprint - but the packaging had a tiny typo: “Viagra” was misspelled as “Vigra.” They reported it. The FDA confirmed it was counterfeit. That person never bought from that site again.What’s Changing in 2026
New tech is making verification easier. Since late 2023, every prescription drug sold in the U.S. must have a unique serial number. You can scan it with your phone using apps like MediSafe. Major manufacturers like Teva and Sandoz now put 2D barcodes on every bottle. These codes link to the drug’s entire history - where it was made, shipped, and stored. By 2025, the FDA plans to launch AI tools that can analyze a pill’s shape, color, and markings using just a photo. Early tests show 99.2% accuracy. But for now, the old-school checks still work best: look at the label, check the pharmacy, verify the lot number, and trust your gut.Final Checklist: Is Your Generic Legit?
- ✅ The pharmacy is licensed and has a .pharmacy domain
- ✅ The prescription was filled by a licensed pharmacist
- ✅ The pill has a clear, consistent imprint (no smudging or fading)
- ✅ The packaging has the manufacturer’s name, lot number, and expiration date
- ✅ The drug is listed in the FDA’s Orange Book
- ❌ No misspellings, foreign text, or crooked labels
- ❌ No pills in plastic bags
- ❌ No website without a prescription requirement
If all these boxes are checked - you’re good. Generic drugs save the U.S. healthcare system over $370 billion a year. They’re not a compromise. They’re a smart choice - as long as they’re real.
Can a generic drug be less effective than the brand-name version?
Legitimate generics are required by law to work the same way as the brand-name drug. The FDA mandates they deliver the same amount of active ingredient into your bloodstream at the same rate. Studies show 98.7% of approved generics meet this standard. If you feel a difference, it’s likely due to inactive ingredients (like fillers) or a psychological effect - not because the drug is weaker. Always talk to your pharmacist or doctor if you notice changes.
Are all generics made in the U.S.?
No. Many generics are made overseas - especially in India and China. But that doesn’t mean they’re unsafe. The FDA inspects over 2,500 manufacturing facilities each year, including foreign ones. As long as the facility is FDA-approved and the drug is listed in the Orange Book, it’s legal and safe. The issue isn’t where it’s made - it’s whether it passed inspection. Always check the manufacturer’s name and lot number.
Why do some generics cost more than others?
Price differences come down to competition. When only one company makes a generic, it can charge more. Once other manufacturers enter the market, prices drop. For example, a 30-day supply of generic lisinopril might cost $4 at one pharmacy and $12 at another. That’s normal. It doesn’t mean one is fake. Always compare prices at different pharmacies - and ask if your insurance covers a cheaper version.
Can I trust generics bought online?
Only if the website is VIPPS-accredited or has a .pharmacy domain. Over 96% of online pharmacies selling drugs without prescriptions are illegal. Many sell counterfeit pills that contain no active ingredient, or worse - toxic substances like rat poison or chalk. The FDA warns that 1 in 10 drugs bought online are fake. Always use a verified pharmacy. If the price seems too good to be true, it probably is.
What should I do if I think I got a fake generic drug?
Stop taking it immediately. Keep the pills, packaging, and receipt. Call your pharmacist and ask them to verify the lot number. Then report it to the FDA’s MedWatch program. You can file a report online at fda.gov/medwatch or by phone at 1-800-FDA-1088. Reporting helps the FDA track fake drugs and protect others. Don’t wait - even one counterfeit pill can be dangerous.








So i just took my generic blood pressure med and it made me dizzy as hell
Called my pharmacy they said its the same stuff
Well i dont care what the science says if my head feels like its floating im switching back to brand
They dont know what its like to live with this
And dont tell me its placebo i felt it in my bones
Oh honey let me paint you a picture
Generic drugs are the pharmaceutical equivalent of buying a knockoff Gucci bag from a guy in an alley who says it's 'authentic'
Yes technically it has the same shape and color but does it have the soul
The craftsmanship
The dignity
And dont even get me started on the fact that half these generics are manufactured in facilities where the QA guy is named Boris and his only qualification is knowing how to spell 'pharmaceutical' backwards
The FDA's bioequivalence standards are robust and backed by decades of clinical data. Over 98% of generics perform identically to brand-name drugs in real-world use. The differences in appearance are purely due to trademark restrictions, not quality. If you're experiencing side effects, consult your pharmacist about inactive ingredients like fillers or dyes - not the active compound. Most concerns stem from confirmation bias or psychological factors, not pharmacological failure.
Did you know the FDA doesn't inspect every single facility overseas? They send inspectors who get shown fake production lines
They're paid by the same companies they're supposed to regulate
And that Orange Book? It's just a list they update when they feel like it
Meanwhile the real drugs - the ones with real science - are being pushed out by lobbyists who own Congress
And don't get me started on how the 2D barcodes are just a distraction while they implant tracking chips in the pills
It is absolutely imperative, nay, non-negotiable, that we address the alarming and deeply concerning phenomenon of pharmaceutical misrepresentation in the modern healthcare ecosystem.
One must not, under any circumstances, assume that the visual dissimilarity of a generic medication equates to its therapeutic inadequacy - however, the emotional and physiological ramifications of perceived inconsistency cannot be trivialized.
Furthermore, the structural integrity of the packaging, the orthographic precision of the labeling, and the veracity of the manufacturer’s identity are all critical indicators of legitimacy - and any deviation, no matter how minute, warrants immediate investigation, documentation, and regulatory escalation.
Moreover, the fact that 78% of counterfeit drugs exhibit misaligned typography is not merely a statistical anomaly - it is a systemic failure of oversight, a breach of public trust, and a moral outrage.
Most generics are fine. If you feel weird, it’s the filler. Talk to your pharmacist. Done.
I really appreciate how thorough this guide is. I’ve been taking generics for years and never knew about the Orange Book. I just checked mine - my amlodipine is listed under Teva. That’s a relief. I also verified my pharmacy’s .pharmacy domain. It’s good to know there are actual tools out there to protect us. Thank you for sharing this.
As a pharmacovigilance specialist with a background in API synthesis, I can confirm that the regulatory frameworks governing generics are indeed robust - but only if the cGMP compliance chain remains unbroken.
Key indicators: batch traceability via UDI, excipient purity profiles (e.g., lactose monohydrate vs. microcrystalline cellulose), and dissolution profile conformity (f2 > 50).
Pro tip: Cross-reference the manufacturer’s FDA 510(k) number with the NDC code on your bottle. If it’s not in the Drug Registration and Listing System (DRLS), it’s not compliant.
It’s amusing how people treat generic drugs like they’re some kind of noble sacrifice - as if choosing a cheaper option is inherently virtuous.
Meanwhile, the real issue is that pharmaceutical companies deliberately design brand-name drugs to be visually distinctive so they can charge 10x more - and now we’re supposed to be grateful that the generic looks different?
It’s not science. It’s corporate theater. And we’re all just actors in their script.
EVERYTHING IS A LIE.
THE FDA IS A SHAM.
THE ORANGE BOOK? A FAKE DATABASE CREATED BY THE PHARMA-CRATIS.
I SAW A VIDEO ON TIKTOK WHERE A MAN SWALLOWED A GENERIC PILL AND HIS STOMACH TURNED BLUE.
THEY’RE USING NANOTECH TO CONTROL US.
AND THE 2D BARCODES? THEY’RE NOT FOR VERIFICATION - THEY’RE FOR GPS TRACKING.
THEY’RE PUTTING MICROCHIPS IN OUR MEDS.
THEY’RE MAKING US DEPENDENT.
AND THEY’RE USING OUR HEALTH DATA TO SELL TO INSURANCE COMPANIES.
YOU THINK YOU’RE SAFE?
YOU’RE NOT.
YOU’RE BEING WATCHED.
REPORT THIS POST.
THEY’RE COMING FOR YOU NEXT.
This is actually super helpful. I’ve been skeptical about generics too - especially after my last refill looked totally different. But now I know to check the lot number and the pharmacy’s website. I even called the manufacturer and they confirmed it was legit. Feels good to have some real tools instead of just guessing. Thanks for the clarity.
India produces 40% of global generics - yet you Americans act like your FDA is some divine institution.
Fact: over 70% of API manufacturing occurs in China and India.
Fact: the FDA inspects less than 3% of foreign facilities annually.
Fact: your ‘approved’ generics often contain 10-15% impurity load.
Do you think your insulin is pure?
Do you think your metformin isn’t laced with industrial-grade solvents?
You are being poisoned - politely - and you are too lazy to ask why.
I just checked my pill and it matches the Orange Book. So relieved. 😊
so i got my generic adderall and it felt like someone poured concrete in my brain
like i wasnt even me anymore
and the pill was a weird blue instead of the green i was used to
so i called my doc and she was like oh that’s just a different maker
well i dont care what the science says i dont feel like myself
and now im scared to take anything
also i think the pharmacy is spying on me
they asked if i wanted to switch to a cheaper one
like… why do they care?
are they trying to kill me slowly?
Let me tell you - this is one of the most important public health guides I’ve read in years.
It’s not just about pills - it’s about autonomy.
It’s about knowing what’s in your body.
It’s about refusing to be passive consumers in a system that treats medicine like a commodity.
Thank you for giving us the tools to fight back - not with anger, but with knowledge.
Every time someone checks a lot number or verifies a .pharmacy domain, they’re not just protecting themselves - they’re protecting their family, their community, and the future of safe healthcare.
Keep doing this work. The world needs more of this.